Normal Mode Analysis Hemochromatosis
Hemochromatosis>>Task 9: Normal mode analysis
Contents
Short task description
Detailed description: Normal mode analysis
Protocol
A protocol with a description of the data acquisition and other scripts used for this task is available here.
General
<figtable id="comparison">
</figtable>
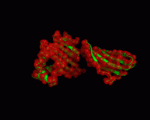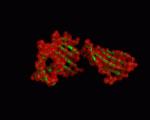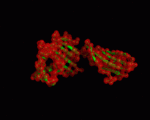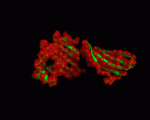
We used the PDB structures 1a6z and 1de4 for our NMA. 1a6z represents HFE bound with Beta-2-Microglobulin only and 1de4 contains the whole TFR-HFE-B2M complex. We extracted HFE's conformations from these structures (chain C from 1a6z and chain A from 1de4). Both files contain the same 272 residues (26-297 within HFE's sequence). A comparison between the two showed that there are only minor differences in the conformation (cf. <xr id="comparison"/>) and almost all of them are within loop regions. TM-Score calculated an RMSD of only 1.602 and a TM-Score of 0.9607 for them.
For the NMA we used WEBnm@ and ElNemo. Both webservers use the C-alpha atoms only for the normal mode calculations and thus could theoretically compute 816 (3N, N = 272) normal modes, though the first 6 are irrelevant as they represent the three simple translations and rotations of the whole protein. In addition to the normal modes they provide also features to compare normal mode motions with other conformations of the same protein or correlation analysis of different normal modes.
WEBnm@
The deformation energy scores computed by WEBnm@ are shown in <xr id="webnma_energy"/>. The first 6 modes (7-12) could be put into three groups: Mode 7-9 which have a very small score, 10 and 11 which have about double the score of 8/9, and Mode 12 which again doubles Mode 10's score. This suggests that the first three combine big motions while the latter three exhibit more and more smaller parts that move separately.
<figtable id="webnma_energy">
| Mode number | Energy score | Mode number | Energy score |
|---|---|---|---|
| 7 | 416.65 | 14 | 4673.81 |
| 8 | 841.18 | 15 | 5862.82 |
| 9 | 849.58 | 16 | 6800.91 |
| 10 | 1436.11 | 17 | 8236.15 |
| 11 | 1899.61 | 18 | 7878.34 |
| 12 | 2981.09 | 19 | 8104.36 |
| 13 | 4518.38 | 20 | 9899.01 |
</figtable>
Modes
<figtable id="webnma_modes7to12">
</figtable>
Correlation of motions
<figure id="correlation">
</figure>
<xr id="correlation"/> shows the correlated motions of all residues within 1a6zC based on the calculated normal modes. At first sight the matrix could be separated into two areas: 1-180 and 181-272. The first one could further be separated into 1-90 and 91-180. When adjusted for the missing residues (1-25) in the PDB file the regions would correspond to 26-115, 116-205, and 206-297 respectively. According to Uniprot these numbers match the residues for HFE's alpha 1 (23-114), alpha 2 (115-205), and alpha 3 (206-297) domains. Though the differentiation in the 1-180 area is not that simple if you are not looking for three domains.
Conformation overlap
<figtable id="webnma_overlap">
</figtable>
We also compared the normal modes' motions with the conformation of HFE in 1de4A to see which of them could be important for the transition between the two conformations. The most notable modes are 18, 29, 30, 52, 72, and 113 (cf. <xr id="webnma_overlap"/>). Their cumulative overlap is 15.2 percent which is significantly above the median of 0.088 percent for all modes. The highest peak (~10 percent) is at mode 6 which is kind of strange as it suggests that a simple rotation would make up the single most notable part of the transition. Though you would think that such a rotation is part of aligning the structures in the first place. Altogehter the first 200 normal modes have a cumulative overlap of 66.8 percent.
It is also not surprising that most of the important modes are rather medium-to-high-frequency as the main differences between the two conformations are in the short loop regions and therefore should be caused by local motions instead of global ones.
Atomic fluctuations
<figtable id="webnma_fluctuations">
</figtable>
The normalized (sum up to 100) atomic fluctuations over all modes in 1a6zC are shown in <xr id="webnma_fluctuations"/>. The regions with the highest displacements are, unsurprisingly, almost exclusively loops. This suggests that the conformation of HFE is pretty fixed and does not change much during the protein's lifetime (cf. also <xr id="comparison"/>).
ElNemo
Modes
<figtable id="elnemo_modes7to12">
</figtable>
Overlap
References
<references/>